|
Aldehydes are widely used and play an essential role in technology and biology. 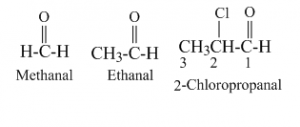
An aldehyde or formal group is a functional group that does not have the 'R' side chain attached. The differences arise because the carbonyl group is inherently polarthat is, the electrons that make up the CO bond are drawn closer to the oxygen than to the carbon. An aldehyde is an organic chemical with the structure C (H)O that contains a functional group. 
The reduction of aldehyde is carried out by using a reagent known as N a B H 4 NaB N a B H 4 than other molecules with different functional groups like ketone, ester, amide, carboxylic acid, as shown in the figure. The aldehyde functional group is a very reactive functional group thus methods to prepare it must be mild and allow the aldehyde group to survive the reaction conditions. The only structural difference between hydrocarbons and aldehydes is the presence in the latter of the carbonyl group, and it is this group that is responsible for the differences in properties, both physical and chemical.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
